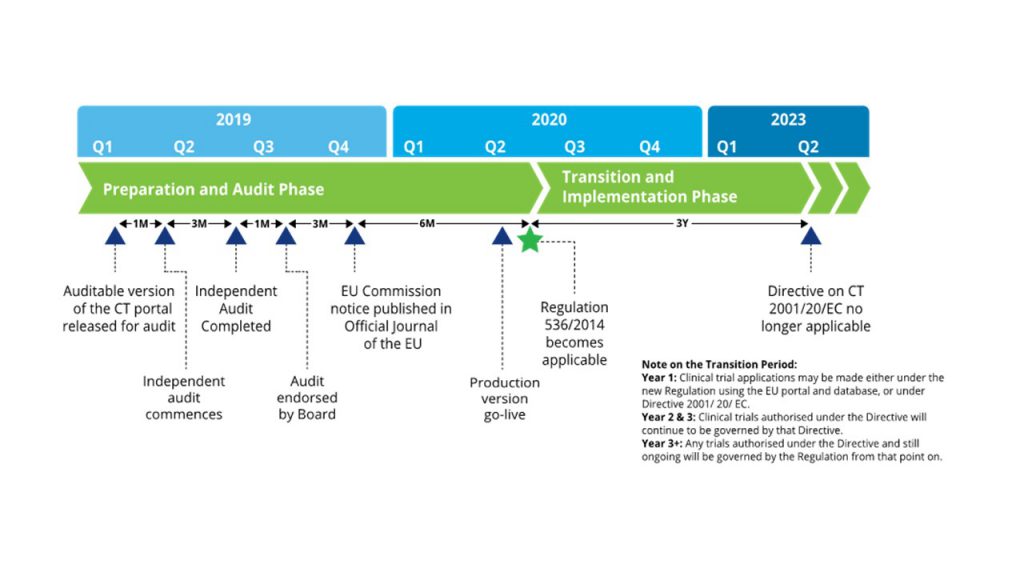
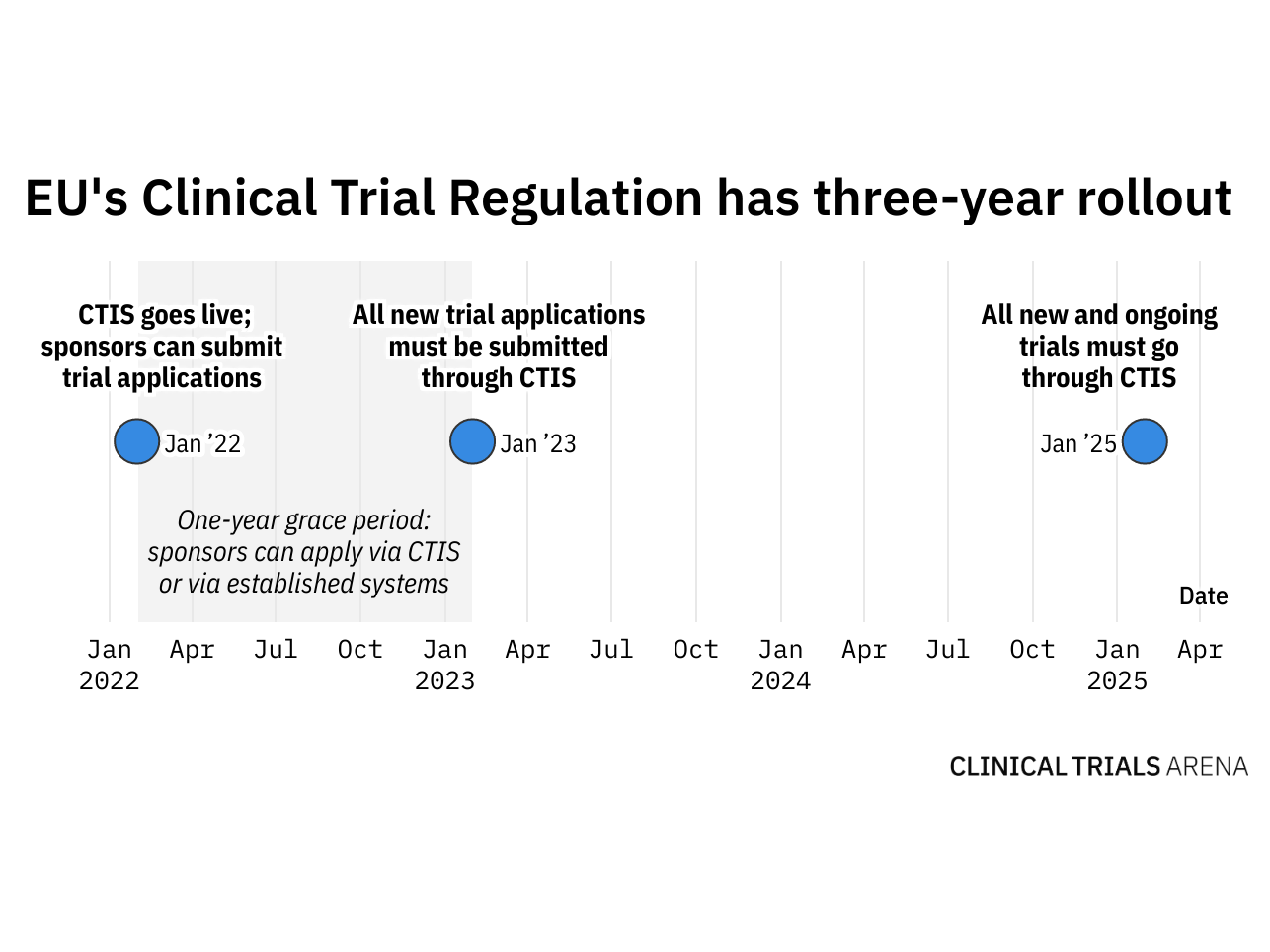
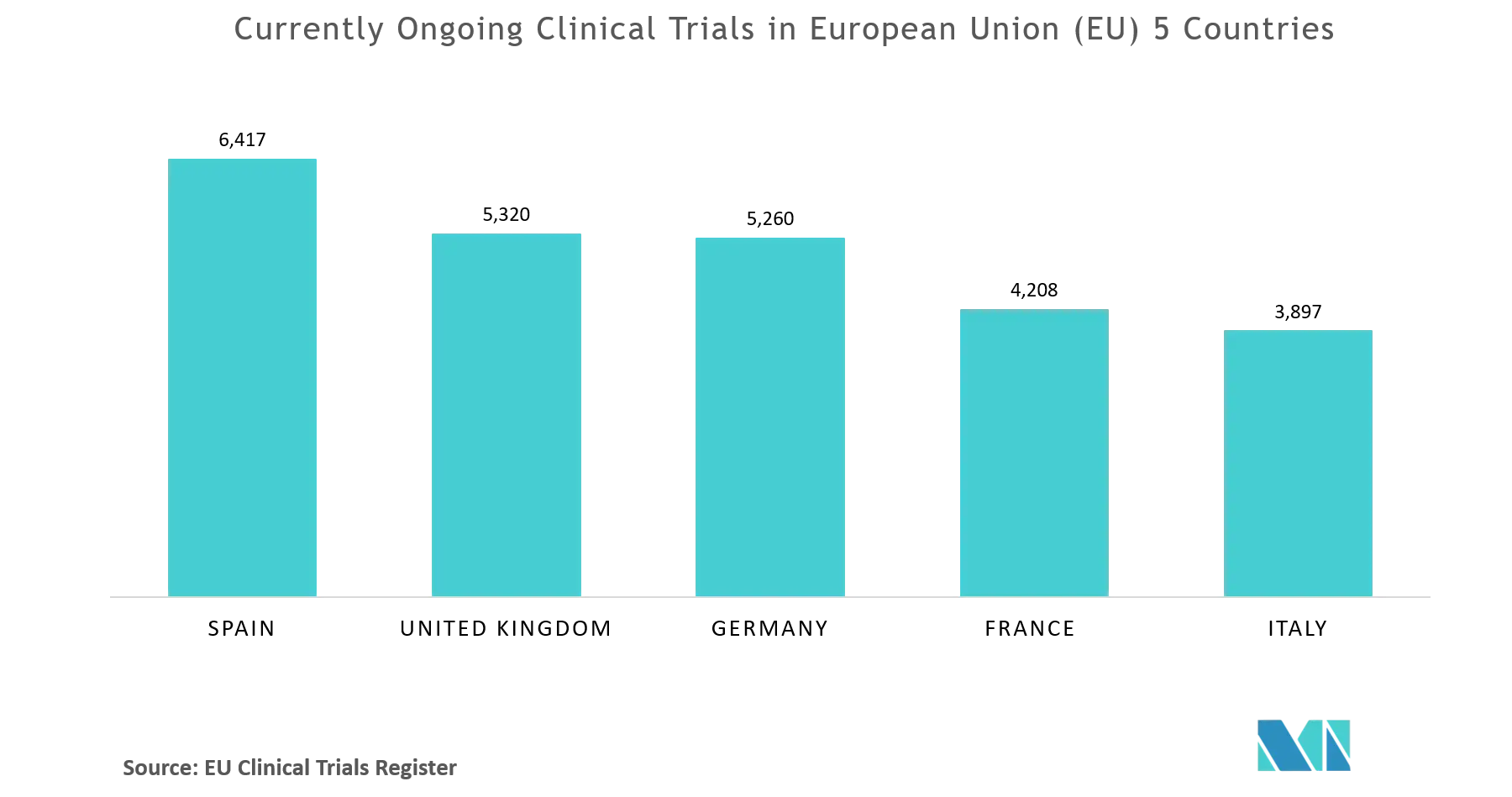
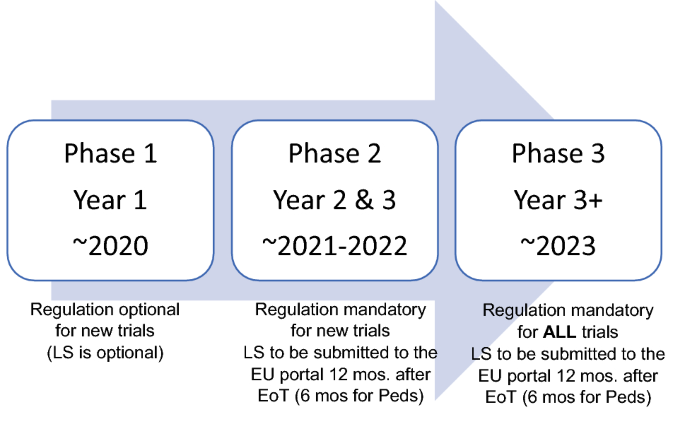
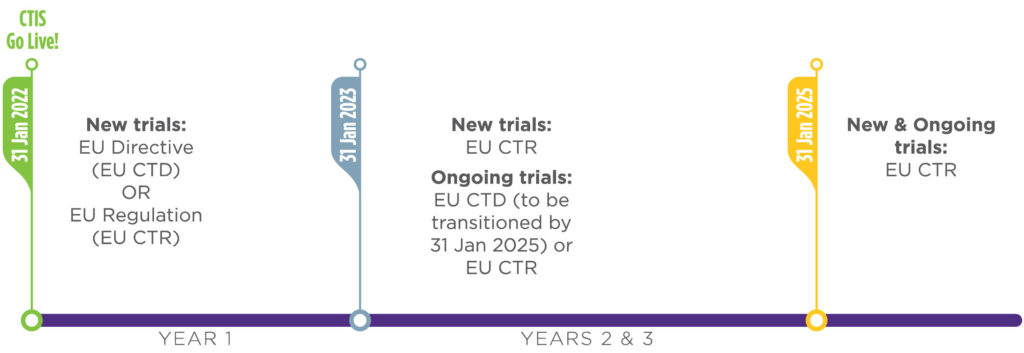
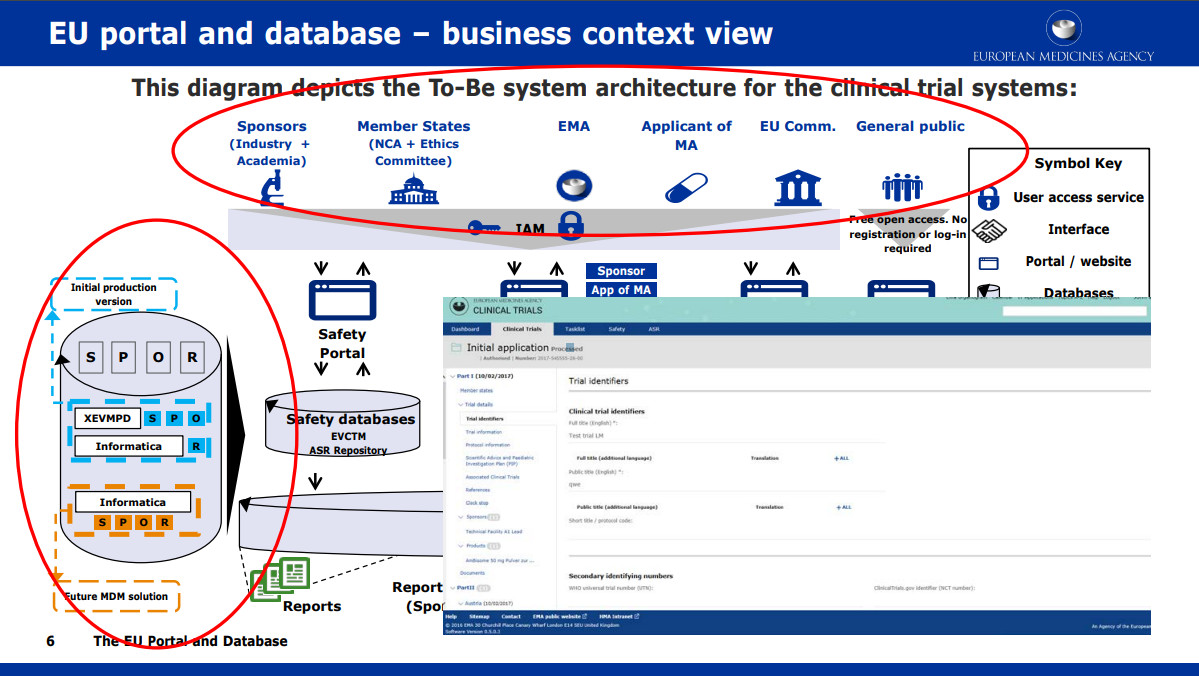
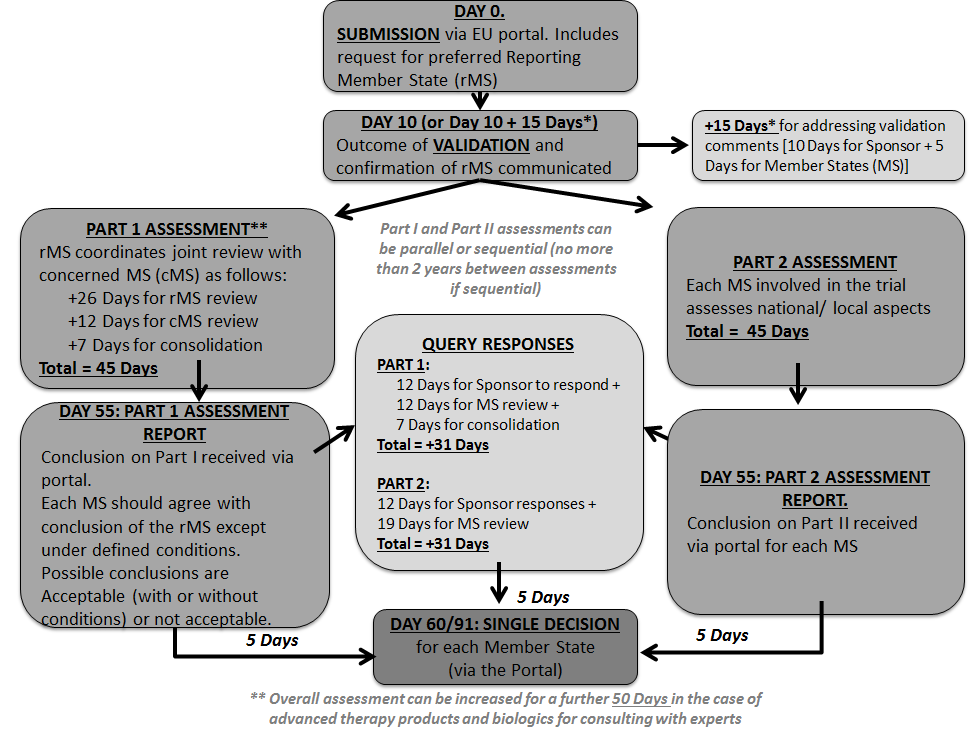
Adapting to the Evolving European Clinical Trial Regulatory Scenario: An Overview of the Current State of the European Clinical Trials Regulation and Clinical Trials Information System - ACRP

Understanding and Implementing the EU Regulation for Initiating and Conducting Clinical Trials - Life Science Training Institute