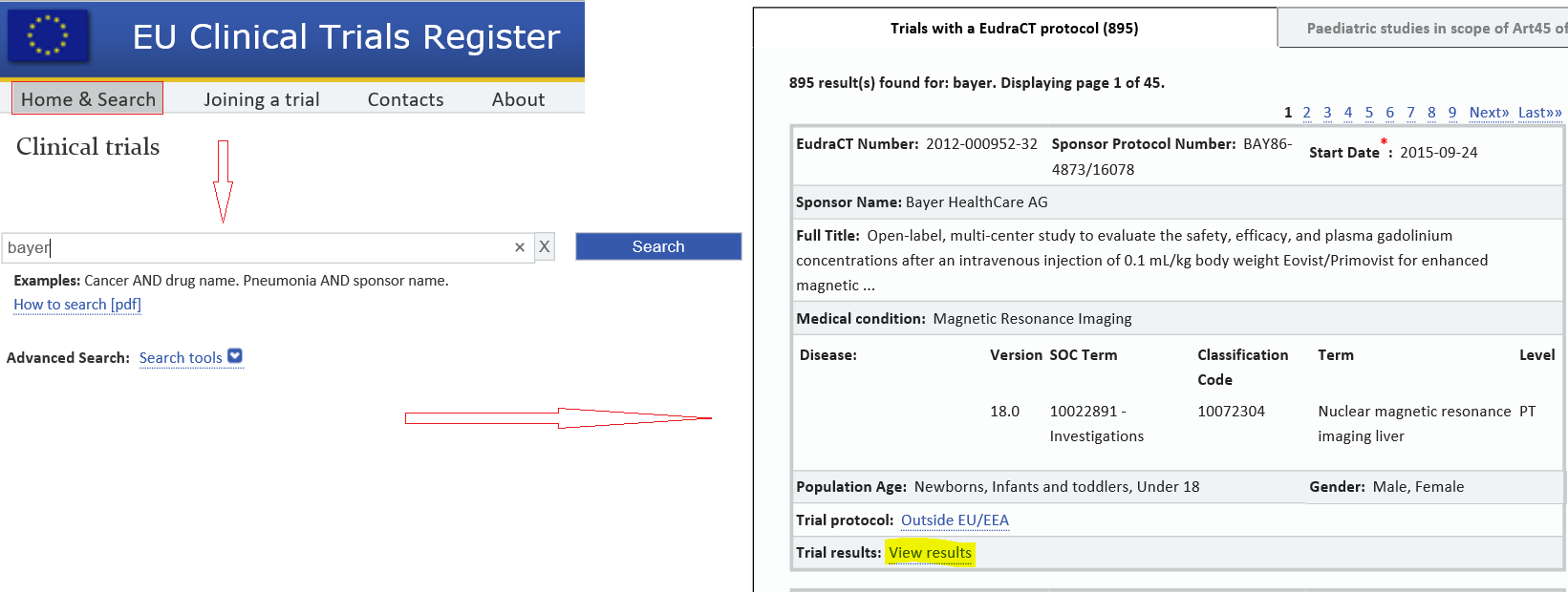
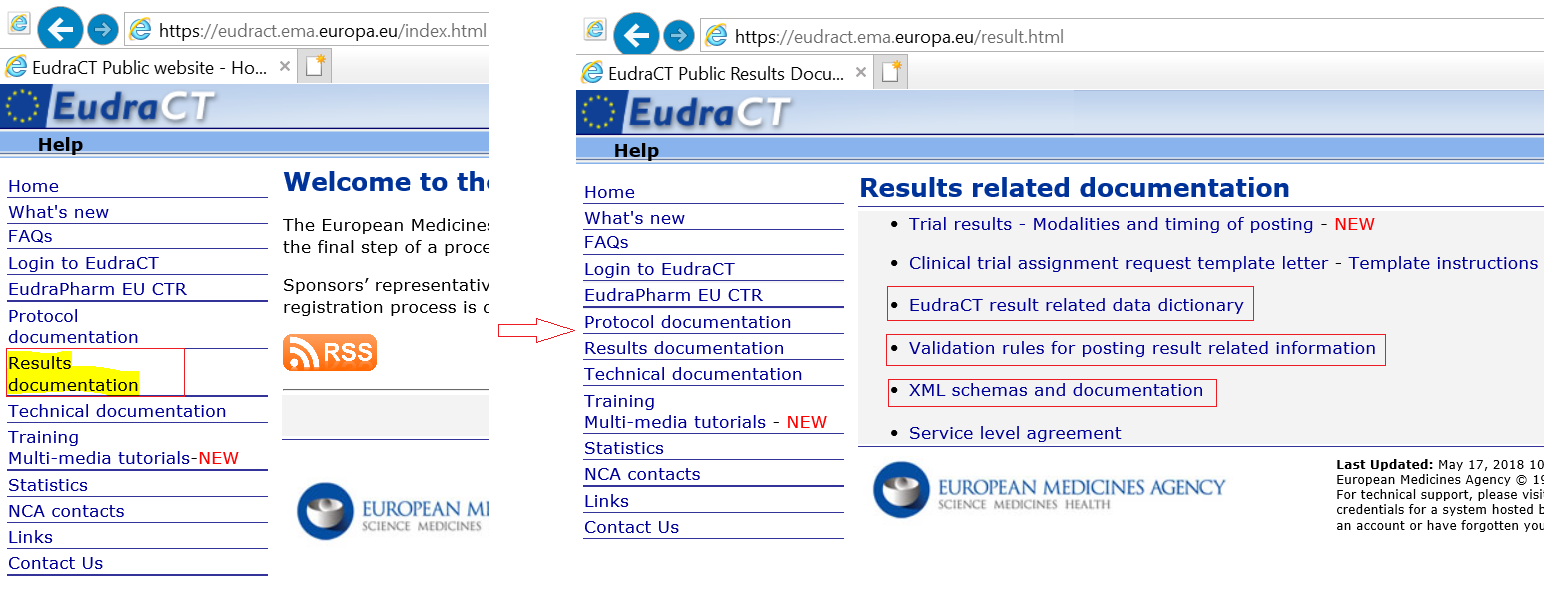
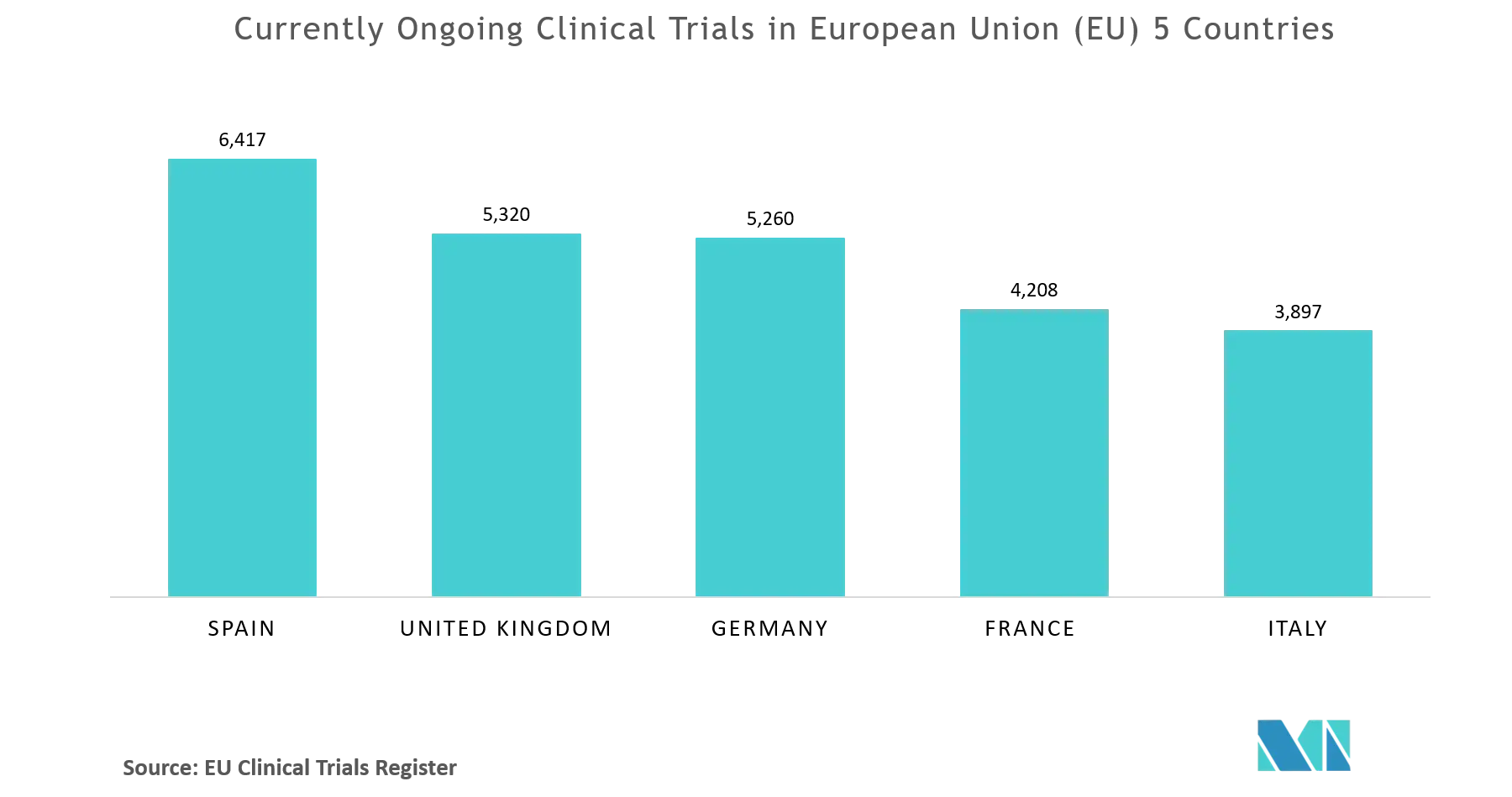
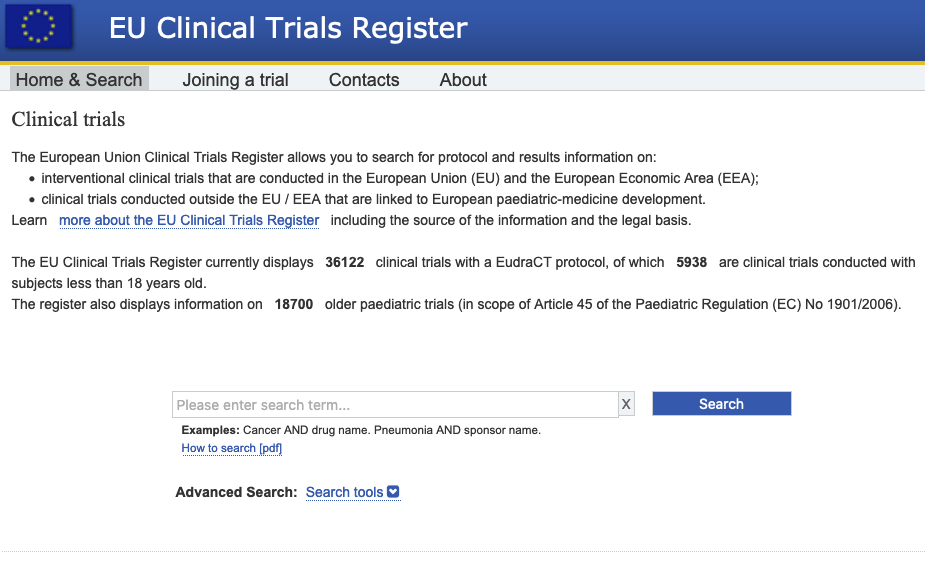
Prevalence of clinical trial status discrepancies: A cross-sectional study of 10,492 trials registered on both ClinicalTrials.gov and the European Union Clinical Trials Register | PLOS ONE

The Current Status of European and National Financial Sources for Clinical Research and Their Impact on Paediatric Non-commercial Clinical Trials: A Case Study of the Czech Republic | SpringerLink

EU Clinical Trial Registry: small industry and academia lack of compliance in reporting Clinical Trial Applications results

Clinical Trial Naming: Using The EU Clinical Trials Register To Create Differentiation - Six Degrees
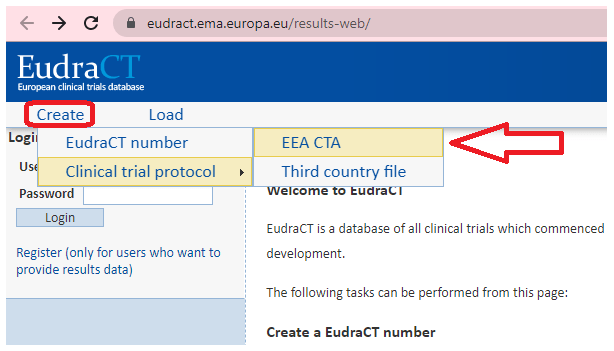
What Are the Documents Required for Clinical Trial Applications to Regulatory Authorities in Europe? - Sofpromed

The Exclusion of Patients with CKD in Prospectively Registered Interventional Trials for COVID-19—a Rapid Review of International Registry Data | American Society of Nephrology