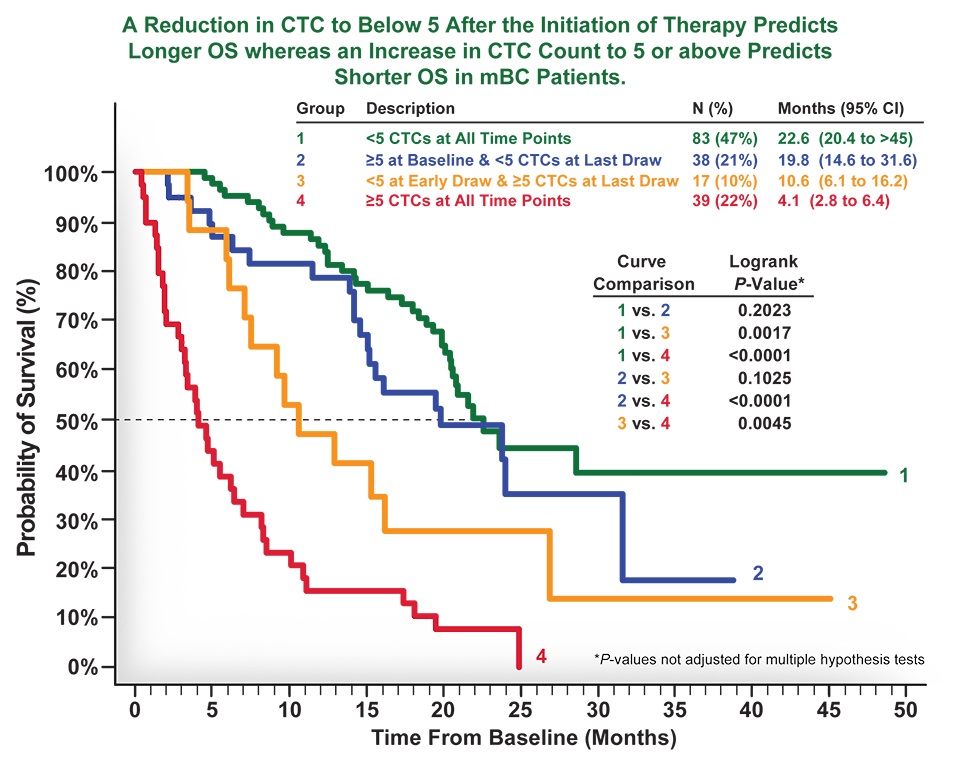
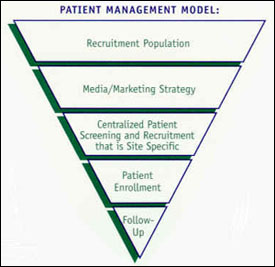
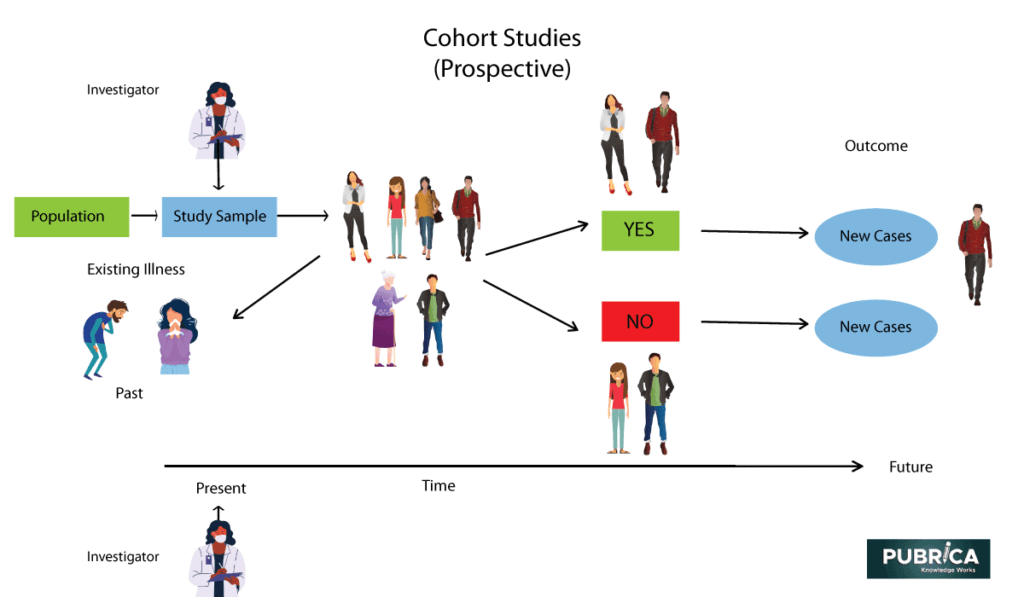
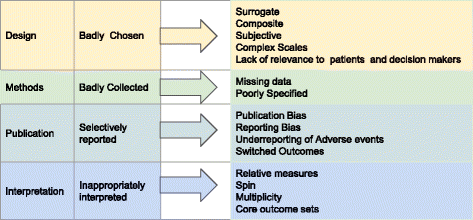
Incorporating Site-less Clinical Trials Into Drug Development: A Framework for Action - Clinical Therapeutics

Optimal design of clinical trials with computer simulation based on results of earlier trials, illustrated with a lipodystrophy trial in HIV patients - ScienceDirect

Schematic of data collection and timing. The initial therapy period for... | Download Scientific Diagram

Radiology workflow for RECIST assessment in clinical trials: Can we reconcile time-efficiency and quality? - European Journal of Radiology

Application of clinical trial inclusion criteria to clinical practice patients to quantify the burden of CNS metastases on health-related quality of life and healthcare resource use in patients with NSCLC - Lung

Follow-up time for each cohort/clinical trial included in the MULTI... | Download Scientific Diagram

Long-term follow-up of a prospective phase 2 clinical trial of extended treatment with rituximab in patients with B cell post-transplant lymphoproliferative disease and validation in real world patients | SpringerLink

EPIC: an evaluation of the psychological impact of early-phase clinical trials in cancer patients - ESMO Open
Guidance on Research Follow-Up Defining a follow-up period is important to the research protocol (even if the follow-up period i
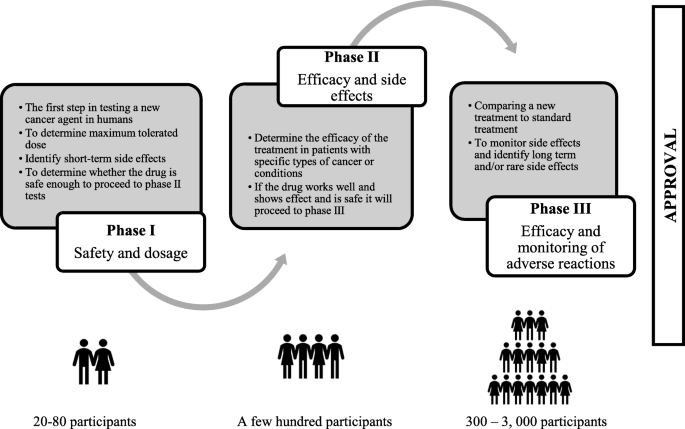
Are cancer patients better off if they participate in clinical trials? A mixed methods study | BMC Cancer | Full Text
Pregnancy Incidence and Correlates in a Clinical Trial Preparedness Study, North West Province South Africa | PLOS ONE