
Randomized Clinical Trial Comparing the Safety and Efficacy of a Hydromorphone Titration Protocol to Usual Care in the Management of Adult Emergency Department Patients With Acute Severe Pain - Annals of Emergency

RADIANCE-HTN: Six-Month Outcomes of a Randomized Trial of Renal Denervation Versus a Sham Procedure for Resistant Hypertension – Impact of Treatment-Blinded Medication Titration | tctmd.com
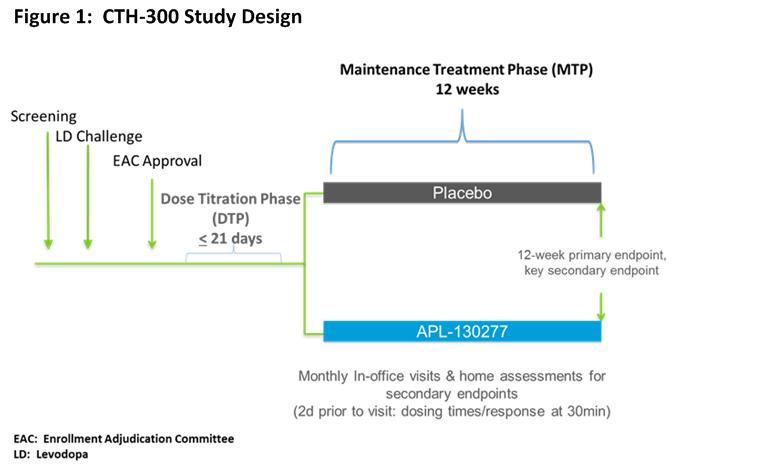
Study Design of an Efficacy and Safety Phase 3, Randomized, Double-Blind, Placebo-Controlled Trial of APL-130277 in the Treatment of OFF Episodes (CTH-300) - MDS Abstracts

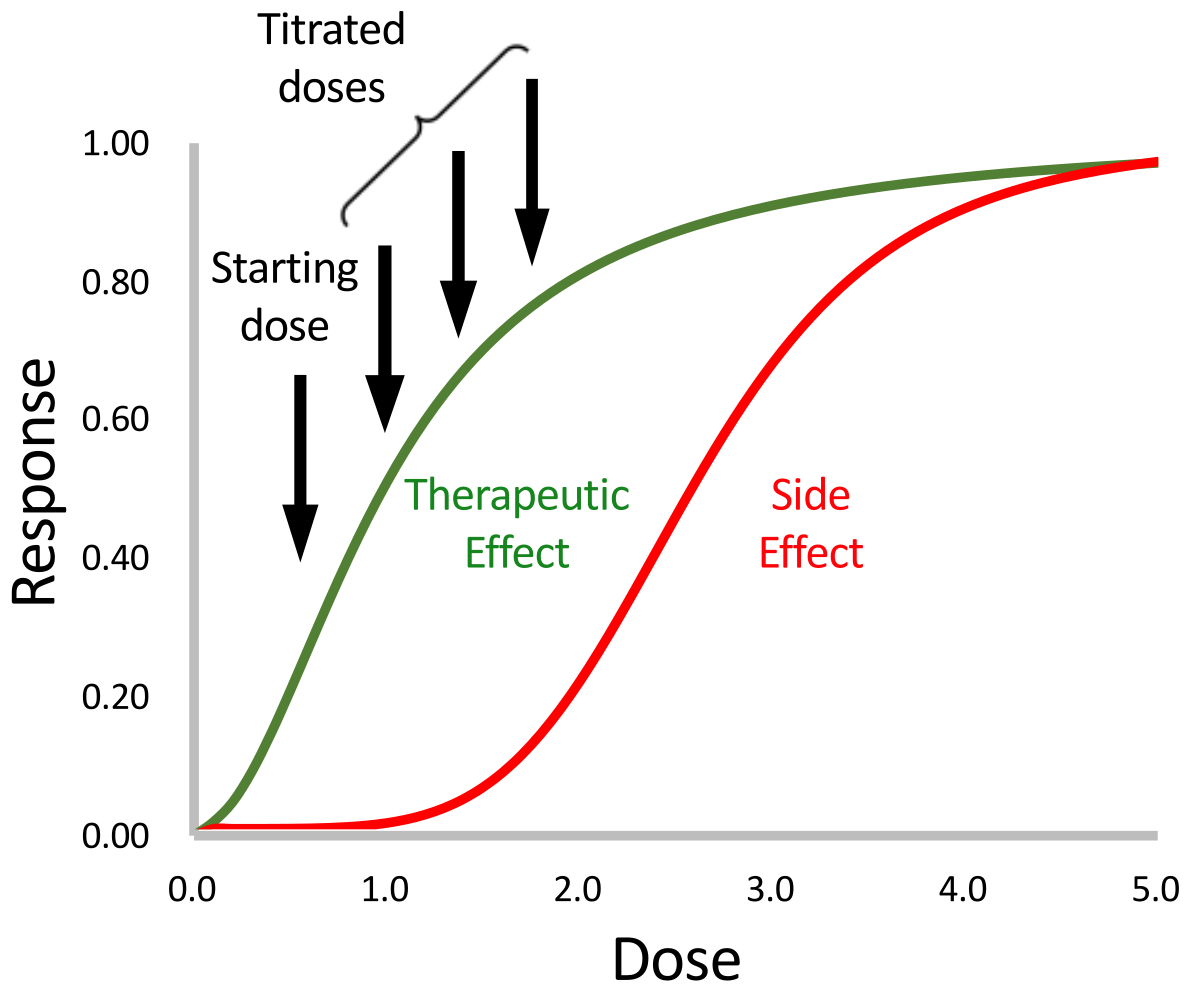
Receiver Operating Characteristic Analysis and Clinical Trial Simulation to Inform Dose Titration Decisions - Clements - 2018 - CPT: Pharmacometrics & Systems Pharmacology - Wiley Online Library

Receiver Operating Characteristic Analysis and Clinical Trial Simulation to Inform Dose Titration Decisions - Clements - 2018 - CPT: Pharmacometrics & Systems Pharmacology - Wiley Online Library



![PDF] Phase 1 trial design: is 3 + 3 the best? | Semantic Scholar PDF] Phase 1 trial design: is 3 + 3 the best? | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/448fbba846c9f38e11aae8ae5d9fab1d28221d3a/5-Figure1-1.png)





:%0A%0APatritumab%20Deruxtecan%20(Up-Titration)%20for%20Lung%20Cancer.png?md=1)